How To Draw Lewis Dot Diagrams
How To Draw Lewis Dot Diagrams - Valence electrons are represented as dots. Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots around it to represent the valence electrons. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. The following is a general procedure for drawing lewis structures. Be sure to leave enough space between the atoms to draw your electrons and bonds. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. Web here are the steps to draw a lewis structure. A video tutorial for how to draw lewis structures in five steps. 2.8m views 10 years ago lewis structures. How to draw a lewis structure. What is the lewis structure for so2? Web creating lewis diagrams is rather simple and requires only a few steps and some accounting of the valence electrons on each atom. And we would account for these valence electrons in our dot structure. Web here are the steps to draw a lewis dot diagram: Web lewis structures made easy: 673k views 5 years ago lewis. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. Web lewis diagrams made easy: Assess the stability of a structure by considering formal charges of atoms. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. Web how to draw lewis structures. Web lewis dot structures. The following is a general procedure for drawing lewis structures. How do you draw the lewis structure for ions? 4.5m views 7 years ago lewis structure tutorial. Draw the lewis dot structure of a given molecule or ion. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. Web lewis dot structures. It will also work with more complex molecules and ions, if you recognize that individual atoms will have the same arrangement of bonds and lone pairs as they do in the simple structures. Enter the formula of the molecule in the field provided for it. Web here are the steps to draw a lewis dot diagram: How do you draw the lewis structure for ionic compounds? The first thing we would need to do is to find the total number of valence electrons. 673k views 5 years ago lewis. Ask me questions on facebook: Write the atomic symbol for each atom. A lewis structure is a diagram that shows the chemical bonds between atoms in a molecule and the valence electrons or lone pairs of electrons. Ionic bonds are formed between charged particles (ions), so an example of an ionic compound would be nacl, whose lewis structure is: Be sure to leave enough space between the atoms to draw your electrons and bonds. Note down a skeletal structure displaying a realistic bonding pattern by means of only the element symbols.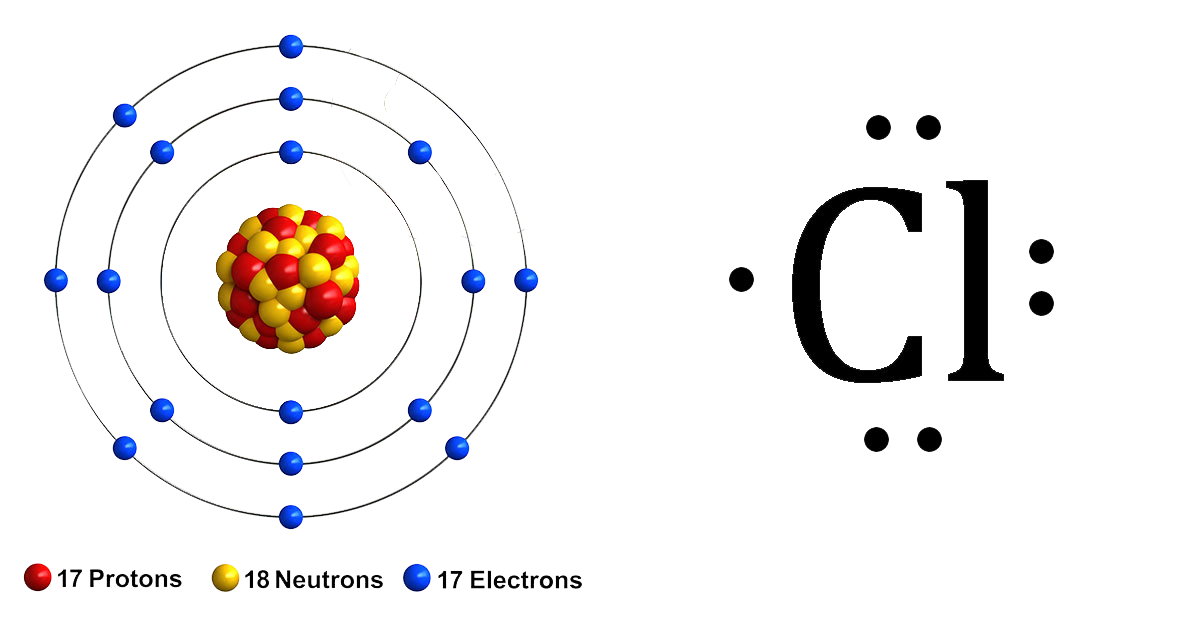
How To Draw Lewis Dot Diagrams Simplereality27

How To Draw Lewis Structures A Step By Step Tutorial

How to Draw a Lewis Structure
Andersen Shows You How To Draw Lewis Dot Diagrams For Atoms.
When Atoms Are Bonded Ionically, The Bond Is Represented By Two Dots Between The Element's Chemical Symbols.
Assess The Stability Of A Structure By Considering Formal Charges Of Atoms.
Using The Periodic Table To Draw Lewis Dot Structures.
Related Post: