How To Draw Dipole
How To Draw Dipole - From in between the hydrogen atoms to the oxygen atom. Web now that we understand how to draw dot structures and we know how to predict the shapes of molecules, let's use those skills to analyze the polarity of molecules, using what's called the dipole moment. Remember, for molecules with one polar bond, the molecular dipole is determined simply. After completing this section, you should be able to. Web it is relatively easy to measure dipole moments: Web two equal and opposite charges separated by some distance constitute a dipole. When two elements have the same electronegativity, there is no dipole. Web how do you determine what forces act when you have big and diverse molecule like an anhydride, e.g. Let's draw the dipoles for all the bonds in carbon. 67 views 6 years ago chemistry guided notes. 9, part 1) jared christensen. Polar molecules increase the charge stored on the plates, and the dipole moment can be obtained (i.e., via the capacitance of the system). Web a dipole moment measures a separation of charge. Parallel to the bond, draw an arrow that points towards the element that is more electronegative. Web this chemistry video tutorial provides a. Web dipole arrows are drawn on lewis structures and point towards the more electronegative atom, since they pull electrons towards them. After completing this section, you should be able to. Polar molecules increase the charge stored on the plates, and the dipole moment can be obtained (i.e., via the capacitance of the system). For a molecule, the overall dipole moment. For one bond, the bond dipole moment is determined by the difference in electronegativity between the two atoms. From in between the hydrogen atoms to the oxygen atom. The net dipole is the measurable, which is called the dipole. Web two equal and opposite charges separated by some distance constitute a dipole. Remember, for molecules with one polar bond, the. This is why in water the dipole arrows are drawn going from hydrogen (low electronegativity) towards oxygen (higher electronegativity). Web now that we understand how to draw dot structures and we know how to predict the shapes of molecules, let's use those skills to analyze the polarity of molecules, using what's called the dipole moment. Just place a substance between charged plates (figure \(\pageindex{2}\)); Remember, for molecules with one polar bond, the molecular dipole is determined simply. We start by looking at a water molecule: Web we have calculated in the previous section that the potential changes linearly in a constant electric field. Looking at the electronegativity and shape of the h2o molecule tells you how the arrow depicts the polarity: The electric field generated by the dipole is proportional to the dipole moment. For a molecule, the overall dipole moment is determined by both the individual bond moments and how these dipoles are arranged in the molecular structure. The net dipole is the measurable, which is called the dipole. Web this organic chemistry video explains how to determine if a molecule is polar and has net dipole moment. Web i believe that within a molecule, you draw dipoles for each bond. The dipole moment \(\overrightarrow p\) is parallel to the line of charge. Identify the type of dipole present. After completing this section, you should be able to. Δpep = qδv = −qeδr = −qed 2cos θ (11.6.2) (11.6.2) δ p e p = q δ v = − q e δ r = − q e d 2 cos.How To Draw Overall Dipole Moment DRAWINGS OF LOVE

How To Draw Electric Dipole Moment DRAWINGS OF LOVE
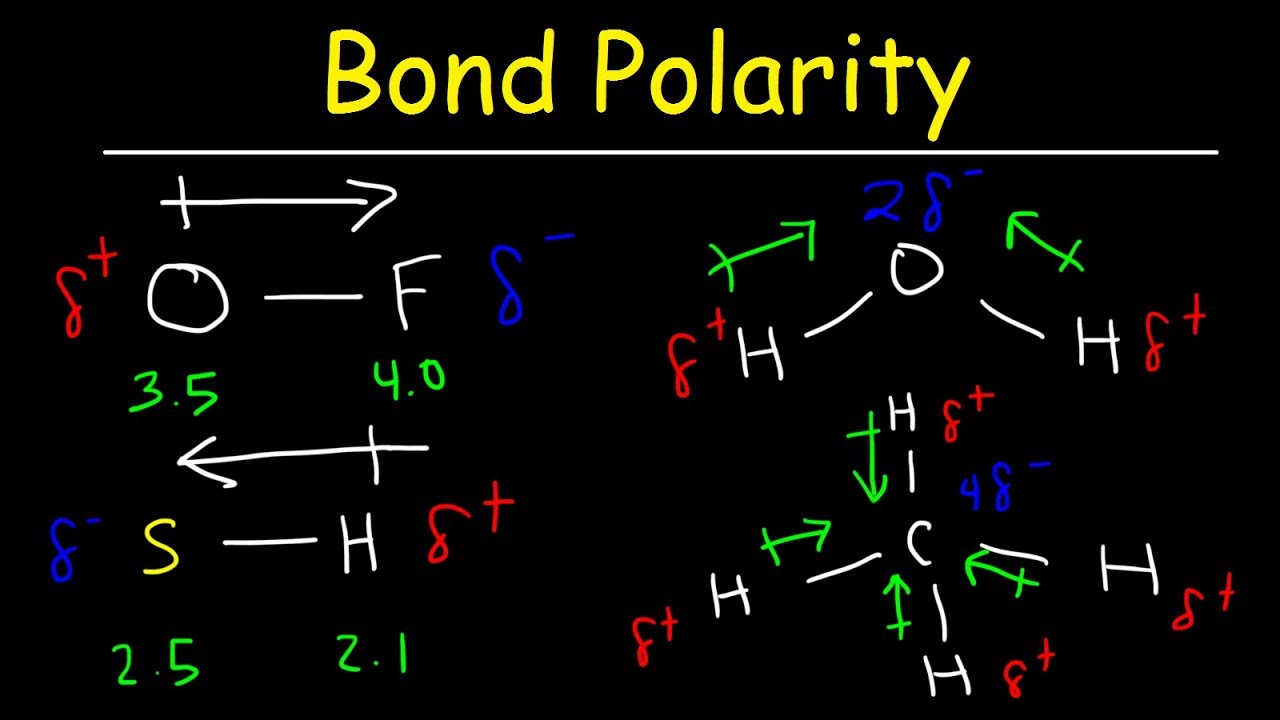
Bond Polarity, Electronegativity and Dipole Moment Chemistry Practice
For One Bond, The Bond Dipole Moment Is Determined By The Difference In Electronegativity Between The Two Atoms.
When Looking At The Bond, You Compare The Two Elements' Electronegativity.
The Difference In Electronegativity Can Be Used To.
From In Between The Hydrogen Atoms To The Oxygen Atom.
Related Post: