How To Draw Bond Dipoles
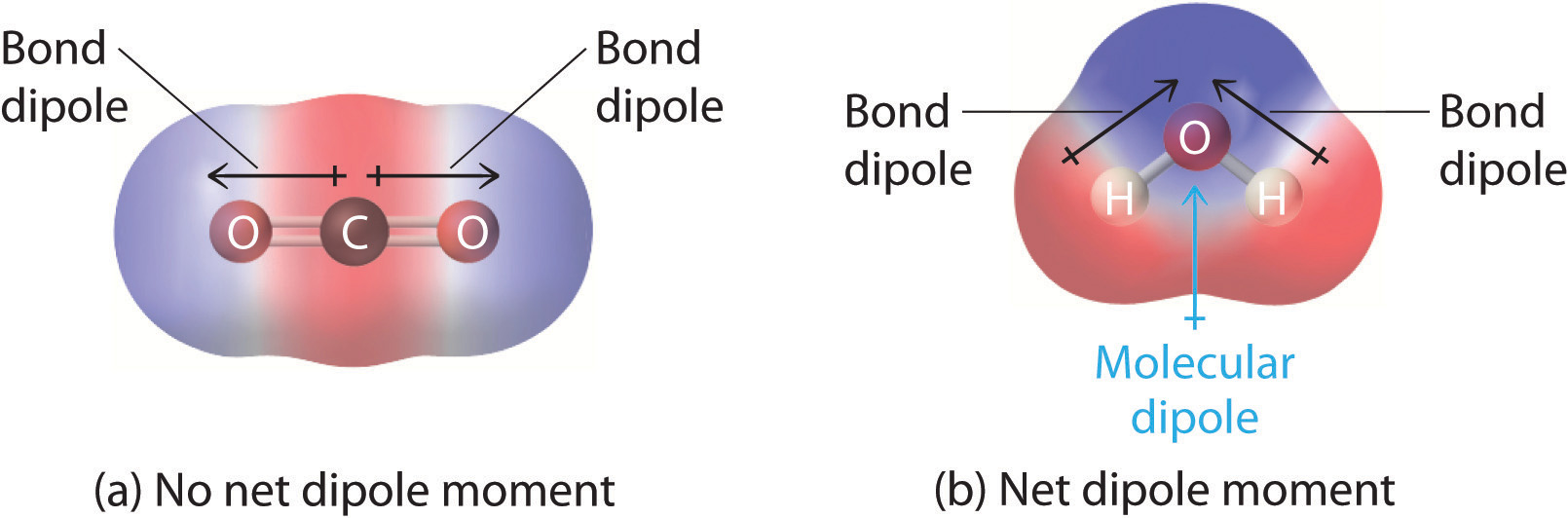
How To Draw Bond Dipoles - Web because of this, the polarization of covalent bonds is typically shown using a special arrow (a dipole arrow) to indicate the direction in which the bond is polarized. Step 4) look at the dipoles. After completing this section, you should be able to. The bond dipole moment that arises in a chemical bond between two atoms of different electronegativities can be expressed as follows: The other atom involved in the bond, the one with the lower electronegativity, would have a partial positive charge, with the plus part of the arrow. They can occur between two ions in an ionic bond or between atoms in a covalent bond; Web we indicate a bond dipole by an arrow with a + at one end and pointing towards the negative end of the bond. Web step 2) draw dipoles for each bond. It explains how to indicate the polarity of a bond and of a. Web after drawing a lewis structure, you'd draw a dipole arrow pointing towards the atom with higher electronegativity. From in between the hydrogen atoms to the oxygen atom. If a substance is both a hydrogen donor and a hydrogen bond acceptor, draw a structure showing the hydrogen bonding. After completing this section, you should be able to. Web dipole vectors are shown as arrows pointing along the bond from the less electronegative atom toward the more electronegative atom.. Web we indicate a bond dipole by an arrow with a + at one end and pointing towards the negative end of the bond. Web step 2) draw dipoles for each bond. The larger the difference in electronegativity, the larger the dipole moment. Web the magnitude of a bond dipole moment is represented by the greek letter mu ( µ). Here, we will learn how to identify molecular dipole moments. Show how the bond dipole moments (and those of any nonbonding pairs of electrons) contribute to the molecular dipole moment. Explain how dipole moments depend on both molecular shape and bond polarity. Using the cross bow arrow shown below we can show that it has a net dipole. You must. Polar molecules increase the charge stored on the plates, and the dipole moment can be. Dipole moments arise from differences in electronegativity. Web dipole moments occur when there is a separation of charge. Bcl 3, for example, has no dipole moment, while nh 3 does. Web the convention in chemistry is that the arrow representing the dipole moment goes from positive to negative. Using the cross bow arrow shown below we can show that it has a net dipole. Example between o and f, the dipole would point to f. From in between the hydrogen atoms to the oxygen atom. Step 4) look at the dipoles. Molecules can be polar or nonpolar, just as bonds can be covalent or polar covalent, depending on the electronegativity values and locations of their atoms. Web the magnitude of a bond dipole moment is represented by the greek letter mu ( µ) and is given by the formula shown here, where q is the magnitude of the partial charges (determined by the electronegativity difference) and r. Web dipole moment (µ) = charge (q) * distance of separation (r) it is measured in debye units denoted by ‘d’. They can occur between two ions in an ionic bond or between atoms in a covalent bond; If δen < 0.5, we usually say that the bond is ionic. You must be able to combine your knowledge of molecular shapes and bond polarities to determine whether or not a given compound will have a dipole moment. Dipole arrows point towards the more electronegative element.
Dipole Dipole Forces of Attraction Intermolecular Forces YouTube

How To Draw Overall Dipole Moment DRAWINGS OF LOVE
Bond Polarity/ Dipoles Chemical Bonding
And In This Case We Have Four Dipoles, But They're Going To Cancel Out In Three Dimensions.
Web This Chemistry Video Tutorial Provides A Basic Introduction Into Bond Polarity, Electronegativity, And The Dipole Moment Of A Bond.
The Vector Points From Positive To Negative, On Both The Molecular (Net) Dipole Moment And.
Conversely, The Presence Or Absence Of A Dipole Moment May Also Give An Important Clue To A Compound’s Structure.
Related Post: