How To Draw Atomic Orbitals
How To Draw Atomic Orbitals - The use of quantum theory provides the best understanding to these topics. Although more complex, these diagrams reveal a more realistic case for bonding, allowing electrons to travel about a molecule, rather than in between one. Web for a given atom, the s orbitals also become higher in energy as n increases because of their increased distance from the nucleus. Web truro school in cornwall. Sketch the shapes of s and p orbitals. Web molecular orbital diagrams are complex, involving two additional orbitals, electronegativity, atomic symmetries and atomic energies. Orbital diagrams must follow 3 rules: We classified the different orbital into shells and sub shells to distinguish them more easily. Make certain that you can define, and use in context, the key terms below. Web © 2024 google llc. Make certain that you can define, and use in context, the key terms below. Web © 2024 google llc. Exploring different orbital shapes and orientations. This organic chemistry video tutorial explains the hybridization of atomic orbitals. Subshells are designated by the letters s , p , d , and f , and each letter indicates a. Orbital diagrams are a visual way to show where the electrons are located within an atom. Sketch the shapes of s and p orbitals. You need to know what type of pi electron contribution each type of. We can break each electron shell down into one or more subshells, which are simply sets of one or more orbitals. In this. Sketch the shapes of s and p orbitals. Web 230313 how to draw shapes of orbitals. List the atomic orbitals from 1 s to 3 d in order of increasing energy. To do so, start with the lowest energy level. 1.9m views 3 years ago new ap & general chemistry video playlist. Electron orbital diagrams and written configurations tell you which orbitals are filled and which are partially filled for any atom. The use of quantum theory provides the best understanding to these topics. It discusses how to determine the. Atomic orbitals and their energies. Web fill each orbital (horizontal line) with the number of occupant electrons. We can break each electron shell down into one or more subshells, which are simply sets of one or more orbitals. Web for a given atom, the s orbitals also become higher in energy as n increases because of their increased distance from the nucleus. Web the goal of this section is to understand the electron orbitals (location of electrons in atoms), their different energies, and other properties. For the p orbitals, draw one arrow pointing up on each of the lines first. Describe the physical significance of an orbital. Let's learn about the shapes of atomic orbitals in this. I will use oxygen ( o2(g)) as an example. After completing this section, you should be able to. Web so, how do these mathematically defined orbitals fit in with the electron shells we saw in the bohr model? Exploring different orbital shapes and orientations. V 1s << v 2s < v 2p.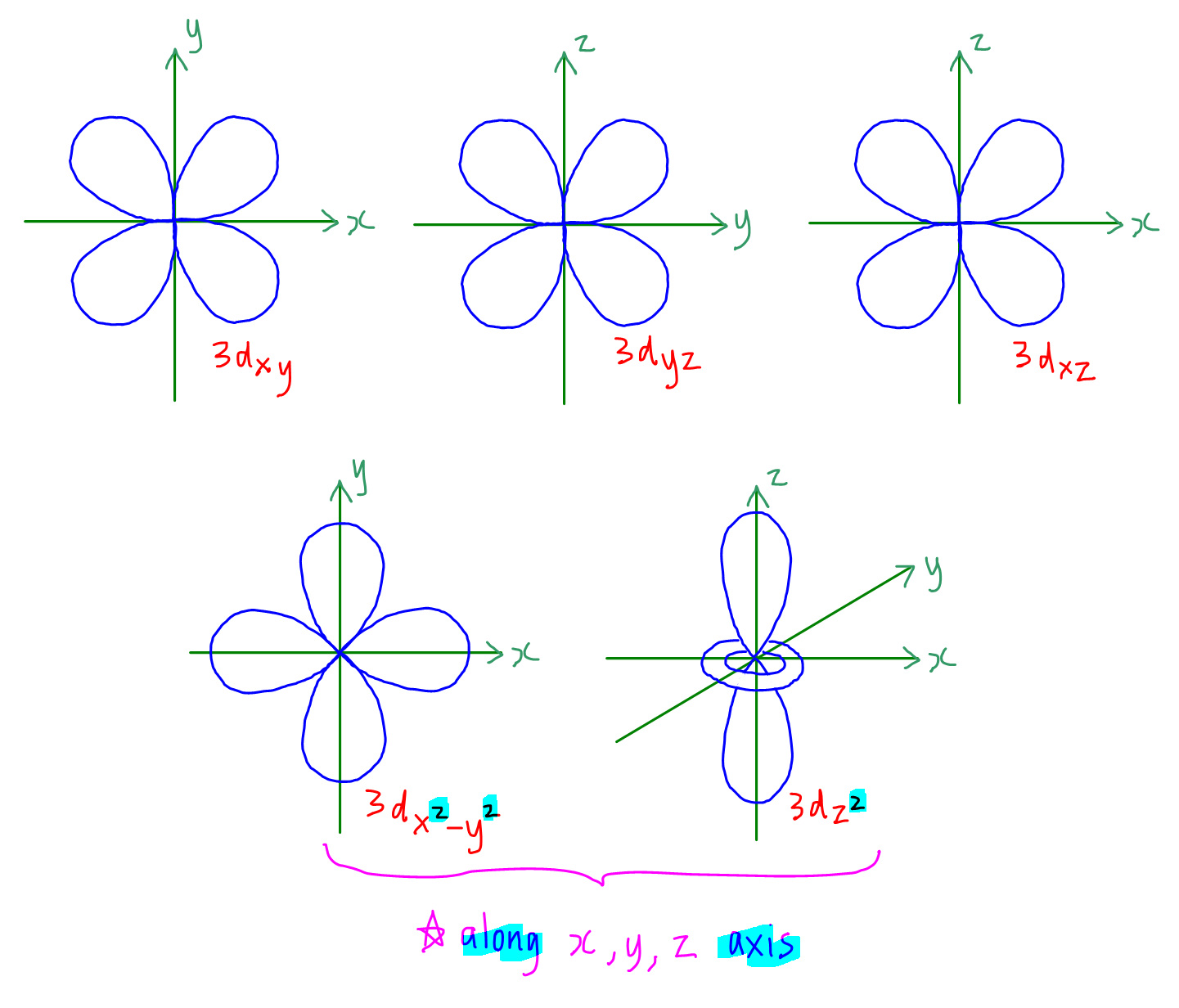
How to Draw Shapes of Orbitals
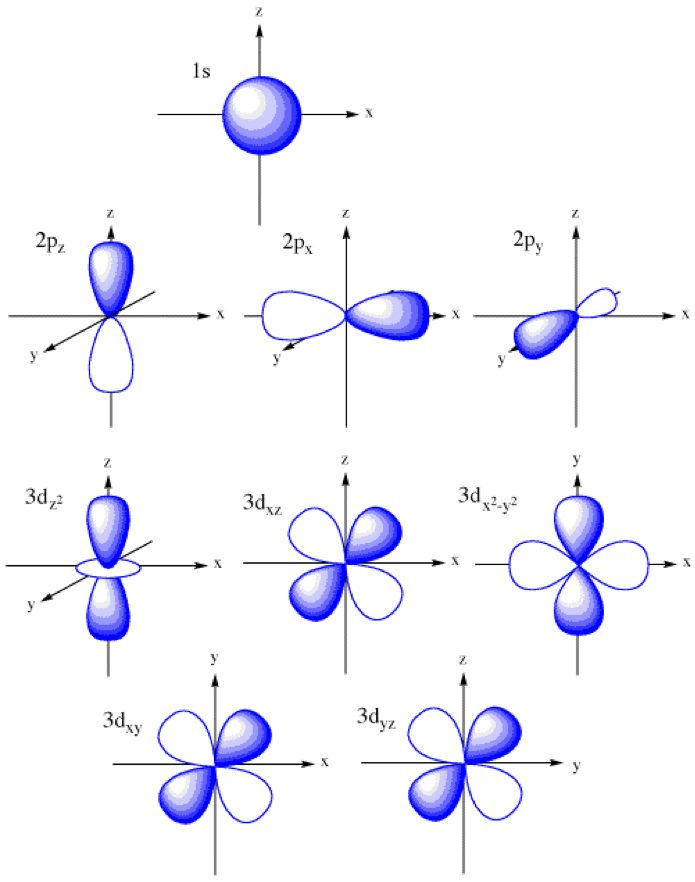
Solved ?1. Consider the s, p, and d atomic orbitals as
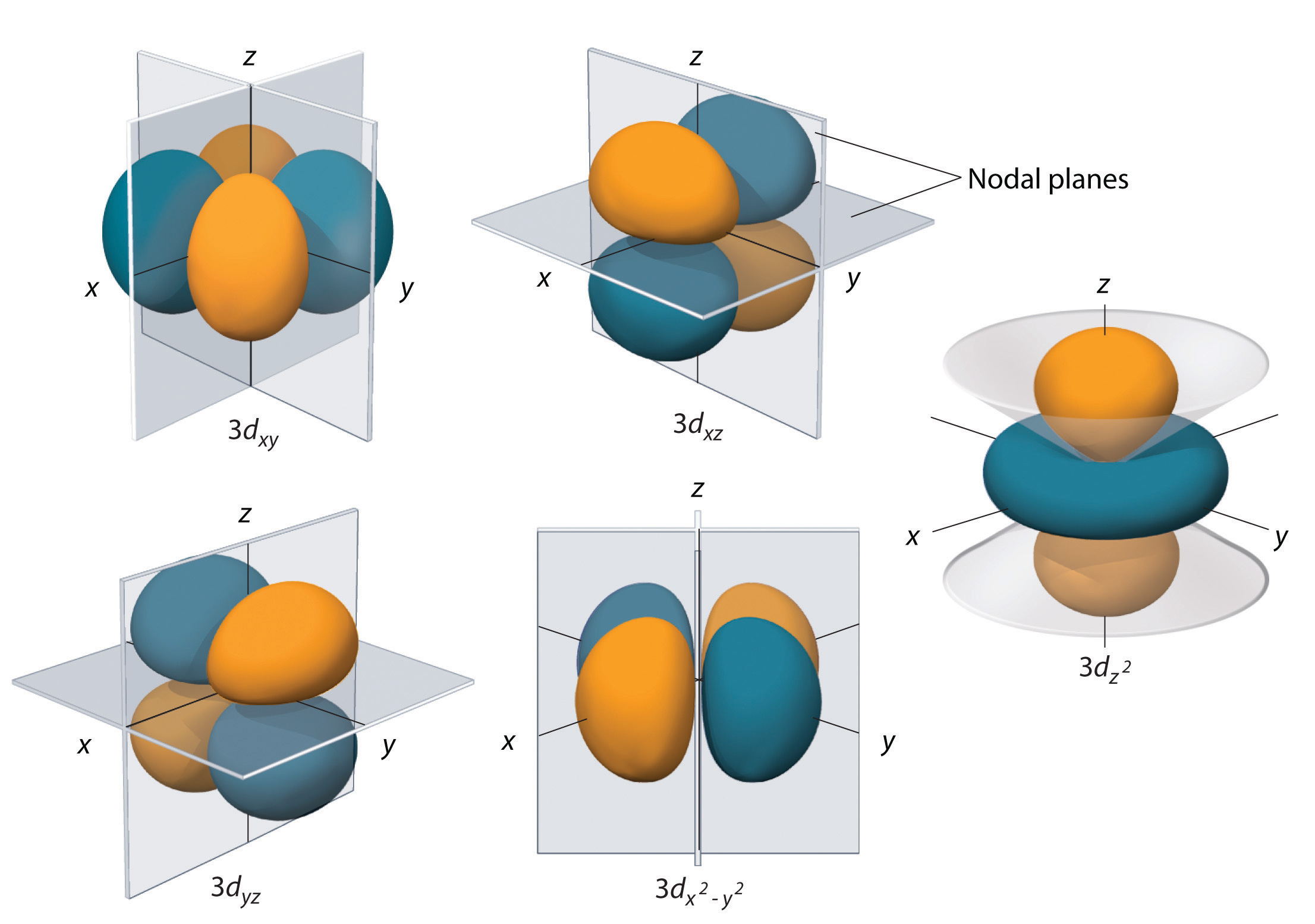
Radial and Angular Parts of Atomic Orbitals Chemistry LibreTexts
To Do So, Start With The Lowest Energy Level.
Web Truro School In Cornwall.
Web © 2024 Google Llc.
It Explores S And P Orbitals In Some Detail, Including Their Shapes And Energies.
Related Post: