How To Draw A Resonance Hybrid
How To Draw A Resonance Hybrid - Contrary to his other lewis structures for nitrate, he didn't add the valence electrons around the oxygen atoms. Click to get pearson+ app download the mobile app. Species such as the acetate ion and benzene are no different from any other. Confirm that no rules of. Get a 10 bullets summary of the topic. We say that the best or actual distribution (most stable) is a resonance hybrid of the contributing lewis representations. After completing this section, you should be able to draw all possible resonance structures for a given species. The real structure is a composite, or resonance hybrid, of the different forms. Web this is the kind of 3d picture that resonance contributors are used to approximate, and once you get some practice you should be able to quickly visualize overlapping 2p z orbitals and delocalized pi electrons whenever you see resonance structures being used. When there is more than one type of atom, keep the least electronegative or metallic atom as the central atom. Oxygen atoms (3*6) = 18. In this text, carboxylate groups will usually be drawn showing only one resonance. When looking at a resonance contributors, we are seeing the exact same molecule or ion depicted in different ways. Web this organic chemistry video tutorial explains how to draw resonance structures, how to identify the major resonance contributor, and how to draw. When drawing these kinds of structures, are the valence electrons included? When looking at a resonance contributors, we are seeing the exact same molecule or ion depicted in different ways. In this text, carboxylate groups will usually be drawn showing only one resonance. This real structure (the resonance hybrid) takes its character from the average of all the individual resonance. 84k views 9 years ago orgo basics. Web when you draw resonance structures in your head, think about what that means for the hybrid, and how the resonance structures would contribute to the overall hybrid. Use curved arrows to represent electron movement. Oxygen atoms (3*6) = 18. Resonance is a mental exercise and method within the valence bond theory of. Web when you draw resonance structures in your head, think about what that means for the hybrid, and how the resonance structures would contribute to the overall hybrid. How to draw a resonance hybrid. Resonance is a mental exercise and method within the valence bond theory of bonding that describes the delocalization of. Calculate the total number of valence electrons from each atom. Web the net sum of valid resonance structures is defined as a resonance hybrid, which represents the overall delocalization of electrons within the molecule. Draw curved arrows to show the movement of electrons. When looking at a resonance contributors, we are seeing the exact same molecule or ion depicted in different ways. Showing 1 of 10 videos. How do you find a molecule’s resonance structures? When drawing these kinds of structures, are the valence electrons included? We say that the best or actual distribution (most stable) is a resonance hybrid of the contributing lewis representations. Some resonance structures are more favorable than others. Web this organic chemistry video tutorial explains how to draw resonance structures, how to identify the major resonance contributor, and how to draw the resonance hybrid. It explains how to draw the resonance structures using curved. Resonance is used to represent all the different ways that identical molecules can distribute electrons. We have learned that lewis structure is a straightforward representation of valence shell electrons in an atom, ion, or molecule.
Resonance Chemistry LibreTexts

Resonance Hybrid of nitrate ion is `` YouTube
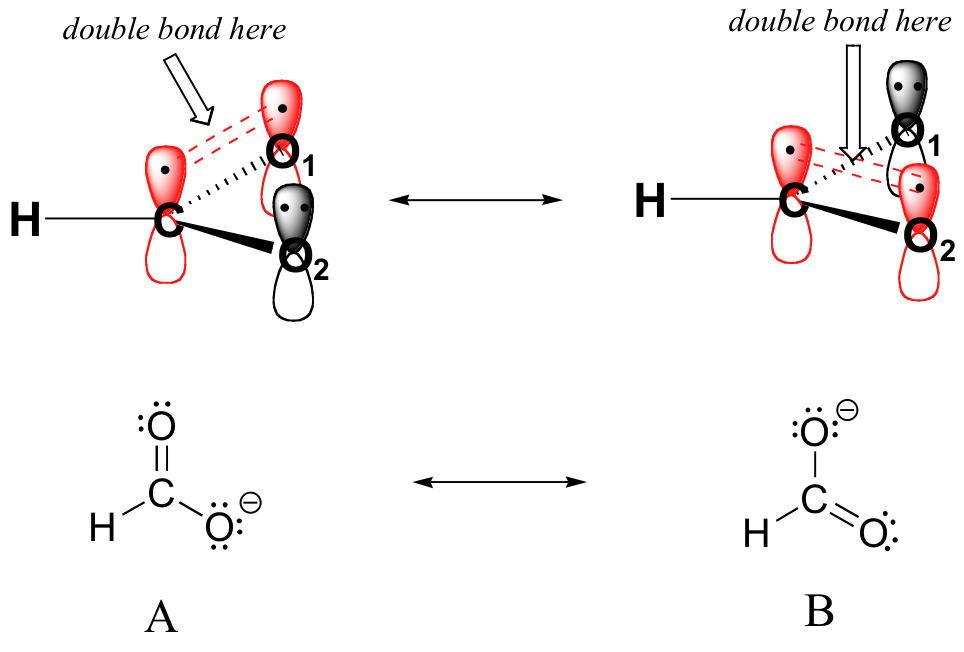
16.4 The Resonance Hybrid Chemistry LibreTexts
The Hybridization (And Hence Molecular Geometry) Assigned To One Resonance Structure Must Be The Same.
Understand The Rules Of Resonance And Identify Where Electrons Can Flow To Or Away From.
Get A 10 Bullets Summary Of The Topic.
Oxygen Atoms (3*6) = 18.
Related Post: