How Do You Draw An Electron Dot Diagram
How Do You Draw An Electron Dot Diagram - So let's say we wanted to draw the dot structure for this molecule, so silicon tetrafluoride. Stages to articulate the electron dot formula are stated beneath. These lewis symbols and lewis structures help visualize the valence electrons of atoms and molecules, whether they exist as lone pairs or within bonds. The sum of the valence electrons is. A beryllium atom, with two valence electrons, has the electron dot diagram below: How many valence electrons does magnesium have? In this video you’ll learn how to draw lewis dot. 234k views 3 years ago. Lewis electron dot diagrams use dots to represent valence electrons around an atomic symbol. A beryllium atom, with two valence electrons, would have the electron dot diagram below. Shared pairs of electrons are drawn as lines between atoms, while. So let's say we wanted to draw the dot structure for this molecule, so silicon tetrafluoride. Atoms and the periodic table. The number of dots equals the number of valence electrons in the atom. Draw a lewis electron dot diagram for an atom or a monatomic ion. Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots around it to represent the valence electrons. Web an electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Since electrons repel each other, the dots for a given atom are distributed. In this video you’ll learn how to draw lewis dot. · 1 · jul 3 2018. A lewis electron dot formula comprises one dot for every valence electron and an element’s symbol. Give examples for molecules and ions that do not follow the octet rule. Determine the total number of valence (outer shell) electrons. A lewis electron dot formula comprises one dot for every valence electron and an element’s symbol. And we would account for these valence electrons in our dot structure. Web lewis structures (also known as lewis dot structures or electron dot structures) are diagrams that represent the valence electrons of atoms within a molecule. These valence electrons are negatively charged and are attracted to the positively charged nucleus, made up of neutrons and protons. The number of dots equals the number of valence electrons in the atom. The sum of the valence electrons is. In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Web lewis structures for covalent molecules: Give examples for molecules and ions that do not follow the octet rule. Then we place the valence electrons around the sides of the box with each side representing an orbital in the outermost energy level. 234k views 3 years ago. · 1 · jul 3 2018. Web you can use a drawing or use small beads or objects to represent electrons and work out the electron arrangement. Draw a lewis electron dot diagram for an atom or a monatomic ion. In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms.
3 Ways to Draw Lewis Dot Structures wikiHow

15 Lewis Dot Structure Examples Robhosking Diagram
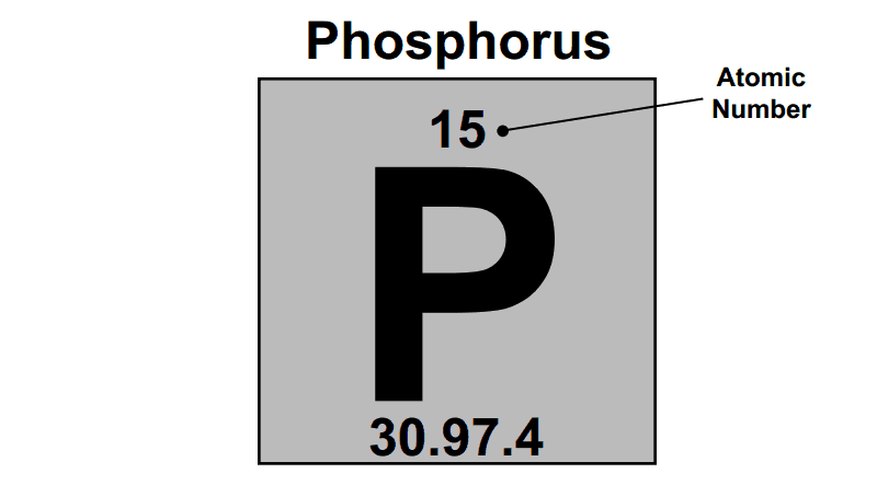
How to Draw Electron Dot Diagrams Sciencing
447K Views 15 Years Ago.
Write The Symbol Of The Atom You Are Drawing The Electron Dot Diagram For In The Middle Of Your Paper.
Download The Following Lesson For Free From Itunes By Typing In The Search Window Papapodcasts.
Move Them Around Until All The Atoms Have 8 In Their Outer Orbits.
Related Post: