Electronegativity Drawing
Electronegativity Drawing - This page deals with electronegativity in an organic chemistry context. Web electronegativity is defined as the ability of an atom in a particular molecule to attract electrons to itself. Web electronegativity is a measure of the tendency of an atom to attract electrons (or electron density) towards itself. It is a dimensionless property because it is only a tendency. In this lesson we will learn (a) how the combination of bonded electrons and lone pairs of electrons result in different molecular shapes and (b) how unequal sharing of. Web electronegativity determines how the shared electrons are distributed between the two atoms in a polar covalent bond. This handout is also included at the end of this packet. Web when is a molecule polar? Electronegativity is a function of: Electronegativity difference is less than 0.4 (nonmetal+nonmetal close together on the periodic table) polar covalent: This handout is also included at the end of this packet. In our previous work we learned why atoms form covalent bonds and how to draw the resulting organization of atoms. If you want a wider view of electronegativity. Web electronegativity is a measure of the tendency of an atom to attract electrons (or electron density) towards itself. Web electronegativity,. See how the molecule behaves in an electric field. Web the electronegativity calculator allows you to calculate the type of bond formed between different elements using their electronegativity values. Web draw the lewis structure; If you want a wider view of electronegativity. Want to join the conversation? This page deals with electronegativity in an organic chemistry context. What does capitol delta look like. Web in this video we look at the concept of electronegativity, how it varies across a period and how to draw lewis dot diagrams for atoms. Web electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract. As shown in figure 2.3 , electronegativities are based on an arbitrary scale, with fluorine the most electronegative (en = 4.0) and cesium the least (en = 0.7). Web follow this procedure for drawing lewis structures until the process becomes second nature. This handout is also included at the end of this packet. The tendency of an atom in a molecule to attract the shared pair of electrons towards itself is known as electronegativity. (b) a is very electronegative, and b and c are not. Web electronegativity is a measure of how easily an atom attracts a pair of electrons to form a chemical bond. In this lesson we will learn (a) how the combination of bonded electrons and lone pairs of electrons result in different molecular shapes and (b) how unequal sharing of. Change the electronegativity of atoms in a molecule to see how it affects polarity. Electronegativity is a function of: In a polar molecule, electron density is unevenly distributed throughout the molecule, resulting in regions of partial negative charge and regions of partial positive charge. Identify the shape of a molecule based on the lewis structure. This activity supports students’ understanding of. See how the molecule behaves in an electric field. A low electronegativity value means an atom readily donates electrons to form a bond or is electropositive. Web in this video we look at the concept of electronegativity, how it varies across a period and how to draw lewis dot diagrams for atoms. The more strongly an atom attracts the electrons in its bonds, the larger its electronegativity.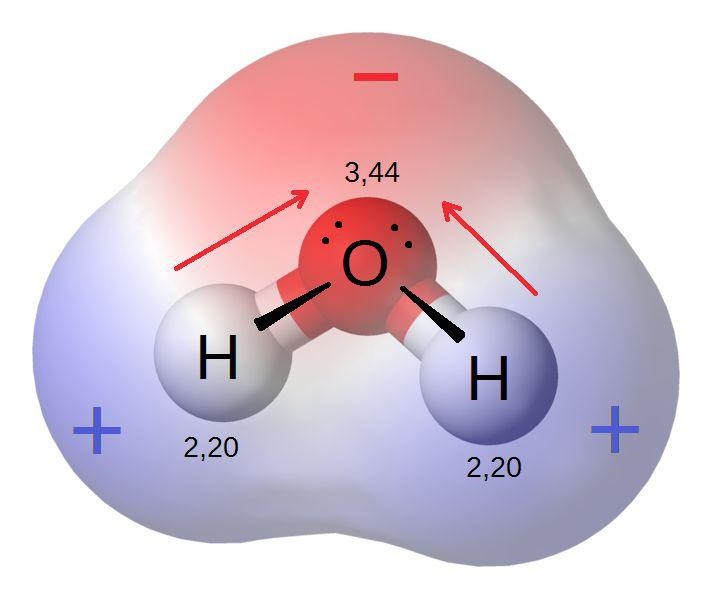
Electronegativity Facts, Summary & Definition Chemistry Revision
Periodic Table of Electronegativities

Electronegativity Definition and Trend
It Explains How To Indicate The Polarity Of A Bond And Of A.
You Can Also Use Our Tool As An Electronegativity Difference Calculator To Determine The Difference Between The Electronegativity Values Of Elements.
Web Draw The Lewis Structure;
Electronegativity Difference In Between 0.4 And 2.0 (Nonmetal + Nonmental Further Apart On The Periodic Table) Ionic:
Related Post:
.PNG)