Draw The Structure Of Acetylsalicylic Acid
Draw The Structure Of Acetylsalicylic Acid - The molecular formula for acetylsalicylic acid is c 9 h 8 o 4 and the expanded formula is ch 3 cooc 6 h 4 cooh. Draw the structure of acetylsalicylic acid and circle the portion of the molecule that shows that it is an ester. The partial lewis structure that follows is for a hydrocarbon molecule. C 9 h 8 o 4. Draw the molecule on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templa nn c 7 h: It is then absorbed into the bloodstream. Its boiling point is 140 ℃. There are 2 steps to solve this one. It is most frequently used to treat mild to moderate pain or to reduce fever. Web structure of acetylsalicylic acid (image will be uploaded soon) properties of aspirin. The carbon—carbon bonds are labeled 1, 2, and 3. Which of the two compounds is a phenol? Acetylsalicylic acid, commonly known as aspirin, is the most widely used drug in the world today. The phenol group on the salicylic acid forms an ester with the carboxyl group on the acetic acid. It is most frequently used to treat mild to. Its boiling point is 140 ℃. Web aspirin is ingested, it is broken down to salicylic acid by the basic conditions in the small intestine. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. (acidic hydrogen atoms are blue.) in comparison, the monoprotic acetylsalicylic acid (asa, aspirin) is less acidic (pka. The carbon—carbon bonds are labeled 1, 2, and 3. Acetylsalicylic acid, commonly known as aspirin, is the most widely used drug in the world today. (acidic hydrogen atoms are blue.) in comparison, the monoprotic acetylsalicylic acid (asa, aspirin) is less acidic (pka = 4.57). Asa is a weak acid that is poorly dissociated in setting the ph of gastric fluid,. Draw the molecule on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templa nn c 7 h: (acidic hydrogen atoms are blue.) in comparison, the monoprotic acetylsalicylic acid (asa, aspirin) is less acidic (pka = 4.57). Acetylsalicylic acid is also not found in nature, similar to many. As a result, the carboxylic groups and the phenyl ring are hybridized. Draw the structure of acetylsalicylic acid and circle the portion of the molecule that shows that it is an ester. There are 2 steps to solve this one. Describe the potential adverse effects of salicylic acid. Web chemistry questions and answers. Which of the two compounds is a phenol? The aspirin molecule contains a total of 21 bond (s). It may be useful to look at each functional group and identify the intermolecular force associated with that functional group. 3g of aspirin can dissolve in 1 liter of water. Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. It is then absorbed into the bloodstream. In the full lewis structure, each carbon atom satisfies the octet rule, and there are no unshared electron pairs in the molecule. Web draw the structures of salicylic acid and acetylsalicylic acid (aspirin) and identify all functional groups.
What is the chemical structure of acetylsalicylic acid? Brainly.in
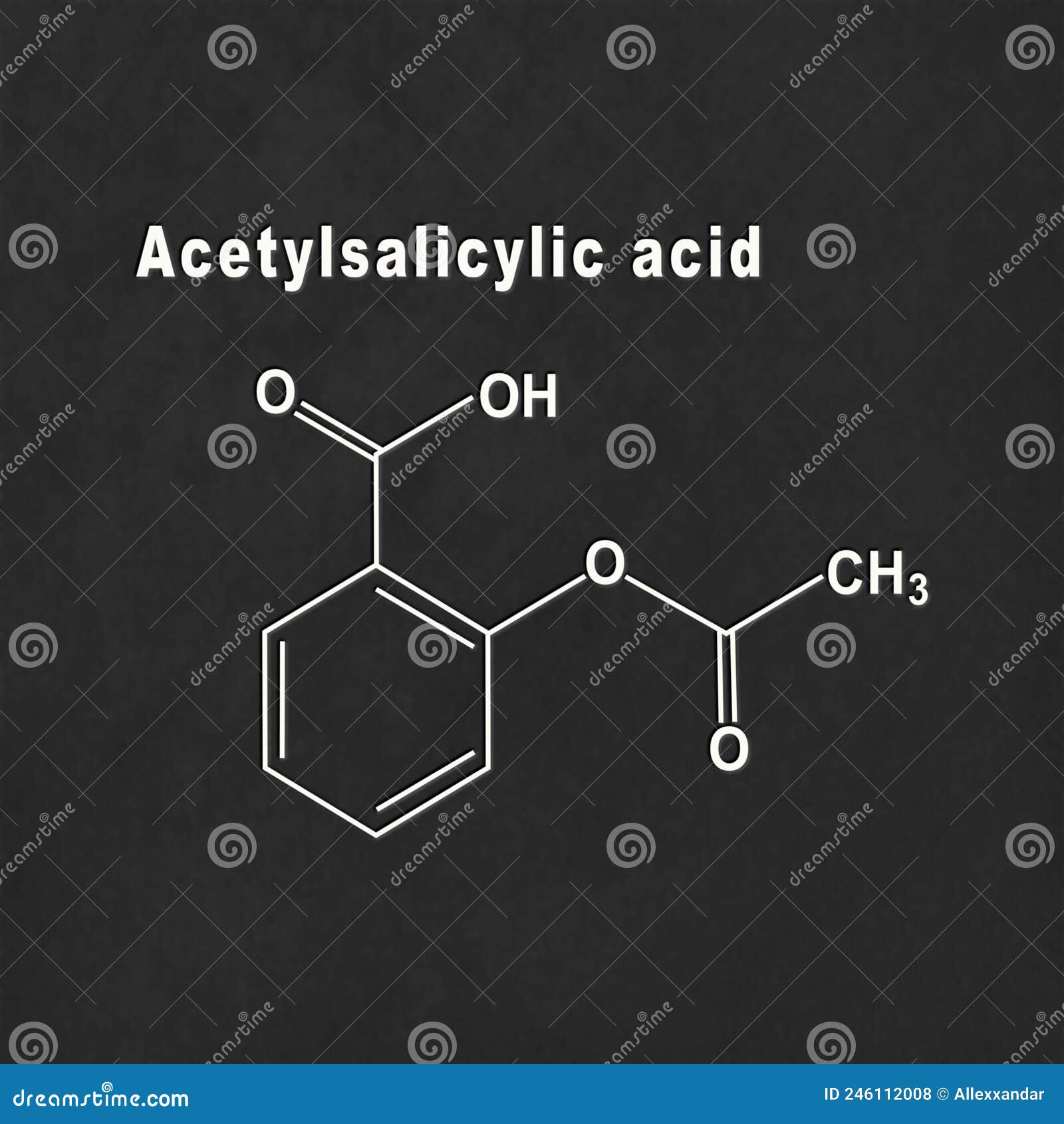
Acetylsalicylic Acid, Aspirin, Structural Chemical Formula Stock
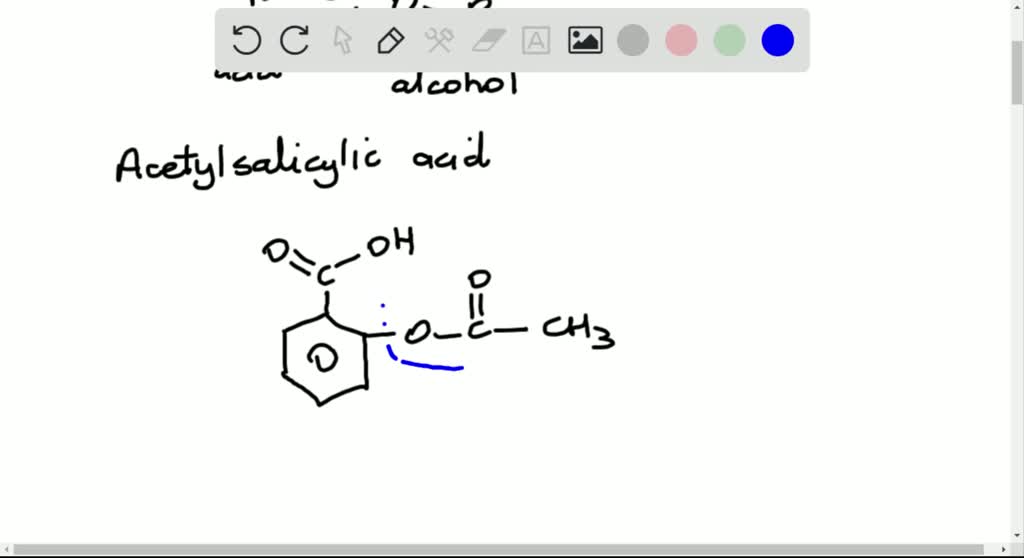
SOLVEDDraw the structure of acetylsalicylic acid, and circle the
From What Acid And Alcohol Is Acetylsalicylic Acid Synthesized?.
This Structure Is Also Available As A 2D Mol File Or As A Computed.
The Molecular Formula For Acetylsalicylic Acid Is C 9 H 8 O 4 And The Expanded Formula Is Ch 3 Cooc 6 H 4 Cooh.
Web According To Its Molecular Geometry, Aspirin Is Planar.
Related Post: