Draw The Orbital Diagram For Oxygen
Draw The Orbital Diagram For Oxygen - It provides insights into the bonding and antibonding interactions between the atomic orbitals of the oxygen atoms. Steps for drawing the molecular orbital (mo) diagram of o2 with its bond order. Web the molecular orbital diagram of o2+ is a representation of the energy levels and bonding between oxygen ions. To illustrate the oxygen orbital diagram, begin by determining the number of electrons from the periodic table. This diagram shows how the electrons in the oxygen atom are arranged in different orbitals. Within orbitals, arrows indicate the spin direction of the occupant electrons. Web electron orbital diagrams are diagrams used to show the energy of electrons within the sublevels of an atom or atoms when used in bonding. Web we can easily draw the mo diagram of o 2 following the steps given below. The video uses kr as an example, but the process is exactly as the same as what you need to do for oxygen. Carbon (atomic number 6) has six electrons. By itself, o2 o 2 is not magnetic, but it is attracted to magnetic fields. Web draw the molecular orbital diagram for the oxygen molecule, o 2. Carbon (atomic number 6) has six electrons. Web the molecular orbital diagram of o2+ is a representation of the energy levels and bonding between oxygen ions. Orbital is the region of space around. By understanding the basics of this diagram, we can gain insights into the stability and reactivity of o2. Web the molecular orbital diagram of o2 is a visual representation of the energy levels and electron configurations of the oxygen molecule. We calculate the bond order as. Steps for drawing the molecular orbital (mo) diagram of o2 with its bond order.. O 2 consists of two oxygen (o) atoms. Web oxygen’s atomic no. Identify and count the pendant atoms' valence orbitals. Molecular orbital diagram for oxygen gas (o2). By understanding the basics of this diagram, we can gain insights into the stability and reactivity of o2. Identify and count the pendant atoms' valence orbitals. Fluorine (atomic number 9) has only one 2 p orbital containing an unpaired electron. How does this diagram account for the paramagnetism of o 2 ? 1s 2 2s 2 2p 4 (for an atom). In this diagram, the oxygen ions are shown as boxes, with the number of electrons and their spin indicated by arrows. From this diagram, calculate the bond order for o 2. Each oxygen atom contributes six electrons, so the diagram appears as shown in [link]. To illustrate the oxygen orbital diagram, begin by determining the number of electrons from the periodic table. Find the point group of the molecule and assign cartesian coordinates so that z is the principal axis. Four of them fill the 1s and 2s orbitals. Oxygen has one more electron than nitrogen and as the orbitals are all half filled the electron must pair up. In writing the electron configuration for oxygen the first two electrons will go in the 1s orbital. Web o2 is usually considered to be double bonded.this means each oxygen atom is sp2 hybridized; Electron configuration through orbit (bohr principle) electron configuration through orbital (aufbau principle) oxygen (o) electron configuration (bohr model) electron configuration. Web if we look at the element after nitrogen in the same period, oxygen (z = 8) its electron configuration is: Web the molecular orbital diagram of o2 is a visual representation of the energy levels and electron configurations of the oxygen molecule.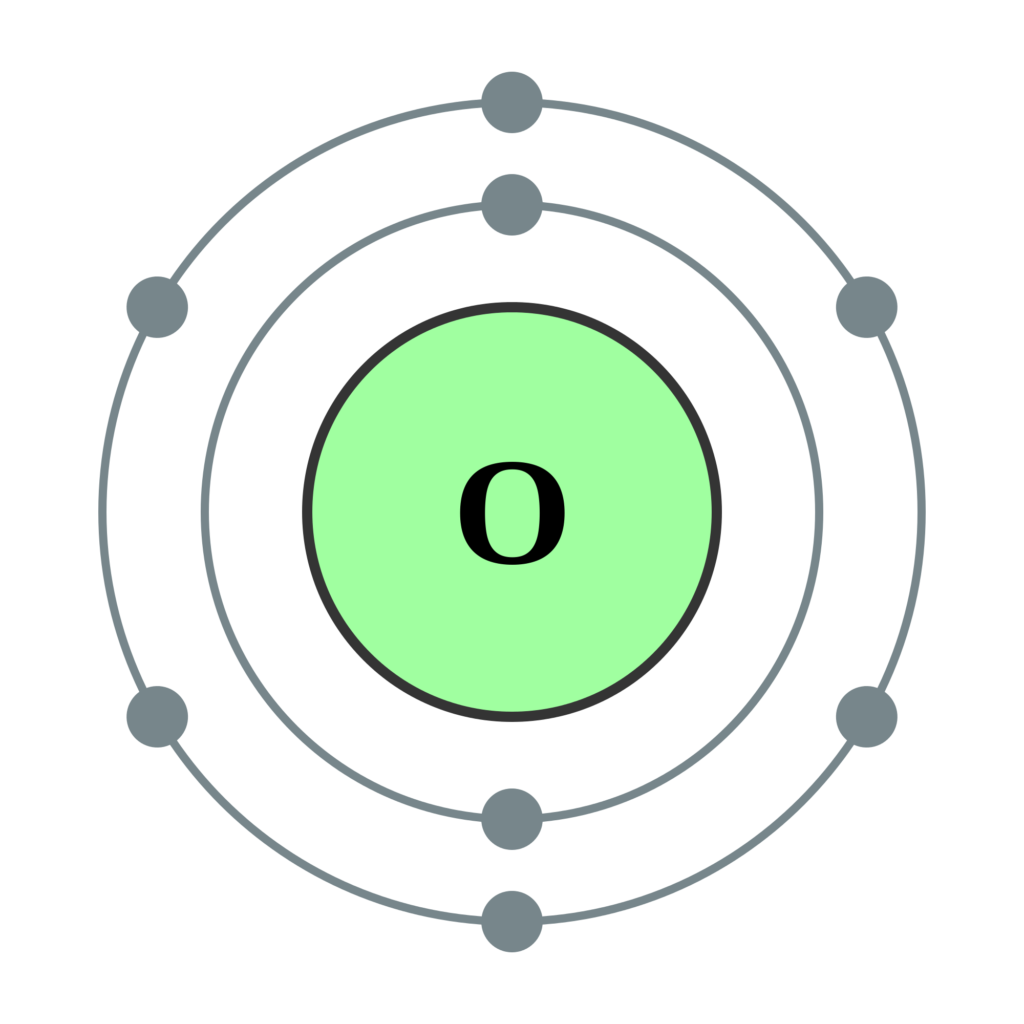
What is the Electron Configuration of Oxygen Archives Dynamic

Molecular Structure of an Oxygen Atom Stock Vector Illustration of

13 Molecular orbital diagram of oxygen molecule. Reproduced from Ref
Web There Is An O=O O = O Double Bond, And Each Oxygen Atom Has Eight Electrons Around It.
Web Oxygen (Atomic Number 8) Has A Pair Of Electrons In Any One Of The 2 P Orbitals (The Electrons Have Opposite Spins) And A Single Electron In Each Of The Other Two.
Web Commonly, The Electron Configuration Is Used To Describe The Orbitals Of An Atom In Its Ground State, But It Can Also Be Used To Represent An Atom That Has Ionized Into A Cation Or Anion By Compensating With The Loss Of Or Gain Of Electrons In Their Subsequent Orbitals.
See The Image Below For Ground State Electron Configuration Of O.
Related Post: