Draw The Lewis Structure Of So3
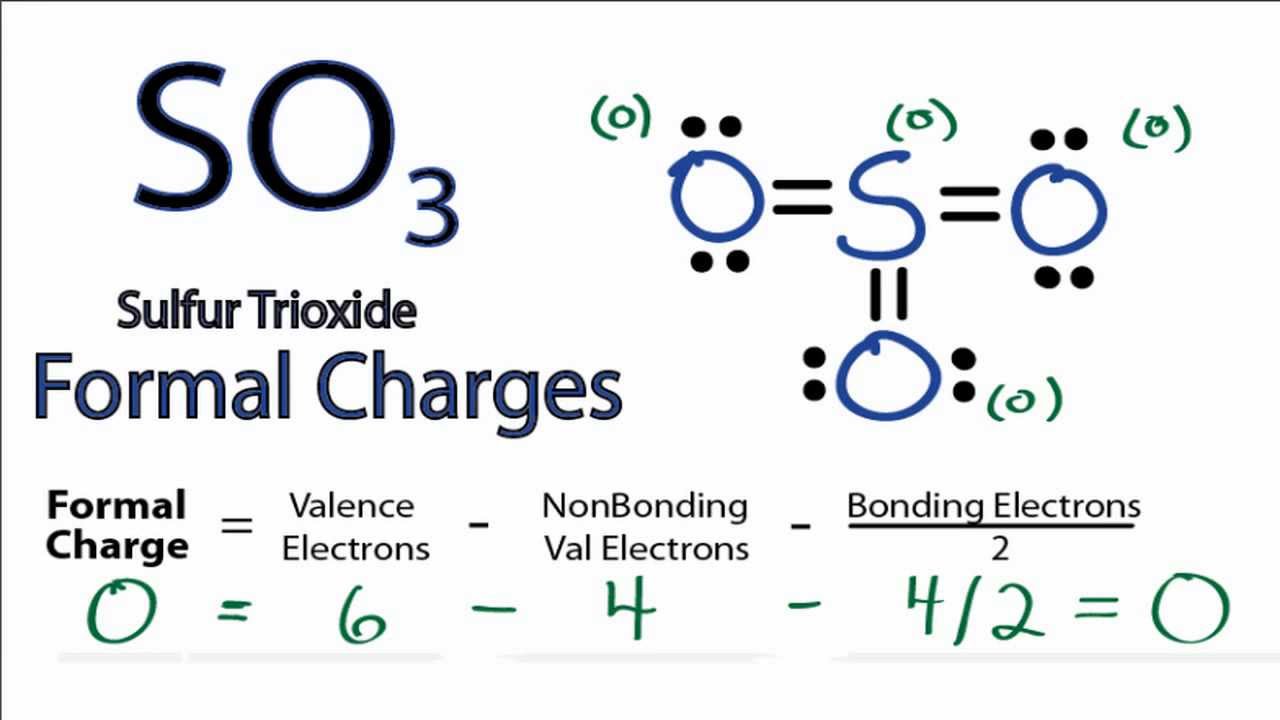
Draw The Lewis Structure Of So3 - When we draw it, firstly we get the three structures at the top. Here, the given molecule is so3 (sulfur trioxide). You might think you've got the correct lewis structure for so 3 at first. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. A lewis structure is a way to show how atoms share electrons when they form a molecule. Determine the total number of valence electrons in the molecule by adding the valence electrons of sulfur and oxygen atoms present in the molecule. This problem has been solved! Lewis structures show all of the valence electrons in an atom or molecule. Drawing the lewis structure for so3. Sulfur brings 6, and oxygen brings 3 each. Send feedback | visit wolfram|alpha. The lewis structure for so 3 is requires you to place more than 8 valence electrons on sulfur (s). Web the so3 lewis structure shows a central sulfur (s) atom with three oxygen (o) atoms around it. Using lewis structures to show valence electrons. Next, look for how many more valence electrons are needed to. 6 + (3 x 6) = 24. Remember, sulfur is in period 3 and can hold more than 8 valence electrons. Be sure to check the formal charges for the lewis structure for so 3. In order to find the total valence electrons in so3 (sulfur trioxide) molecule, first of all you should know the valence electrons present in sulfur. Find more chemistry widgets in wolfram|alpha. This widget gets the lewis structure of chemical compounds. Get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Send feedback | visit wolfram|alpha. Find the total valence electrons in so3 molecule. Draw the lewis structure for the sulfur trioxide (so_3) molecule. Write lewis structures for the following: It discusses the molecular geometry, bond angle, hybridization, and formal charges of the so3. Find more chemistry widgets in wolfram|alpha. Web the so3 lewis structure shows a central sulfur (s) atom with three oxygen (o) atoms around it. Send feedback | visit wolfram|alpha. A lewis structure is a way to show how atoms share electrons when they form a molecule. The initial step in sketching the so 3 lewis structure is to determine the total number of valence electrons. When we draw it, firstly we get the three structures at the top. Web following steps are the main guidelines we have to use for drawing the lewis structure of so 3. Remember, sulfur is in period 3 and can hold more than 8 valence electrons. So 3 is named sulfur trioxide. Both sulfur and oxygen belong to group 16 of the periodic table, resulting in six valence electrons for each of them. Find the total valence electrons in so3 molecule. (please note, none of the solutions are using the expanded octet rule or formal charges) h 2. Web steps of drawing so3 lewis structure.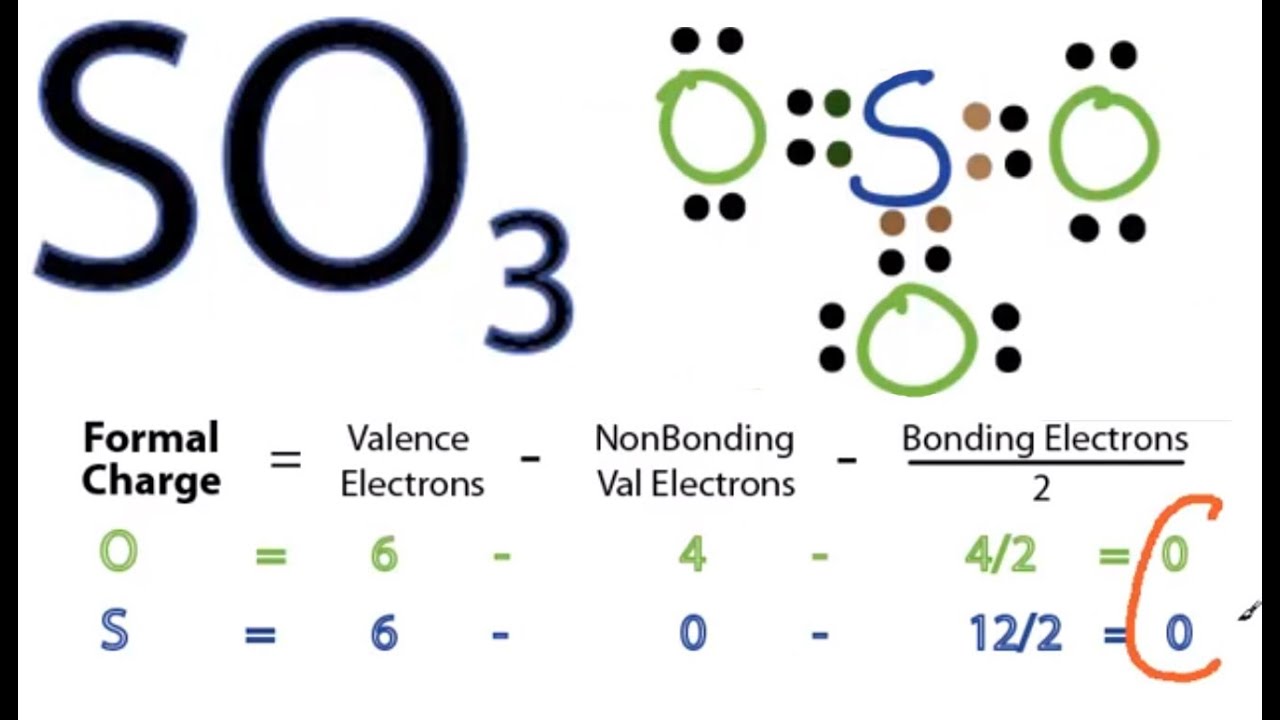
SO3 Lewis Structure How to Draw the Lewis Structure for SO3 (Sulfur

SO3 Lewis StructureLewis structure of SO3 (Sulfur trioxide) YouTube
Draw The Lewis Dot Structure For So3 2 slidesharedocs
Be Sure To Include All Resonance Structures That Satisfy The Octet Rule.
There Are 32 Valence Electrons Available For The Lewis Structure For So 3.
In Order To Draw The Lewis Structure Of So3, First Of All You Have To Find The Total Number Of Valence Electrons Present In The So3 Molecule.
Here, Sulfur In The Center Because Of Its Lowest Electron Capability, And Three Oxygen Around It.
Related Post: