Draw The Lewis Structure For The Xenon Tetrafluoride Molecule
Draw The Lewis Structure For The Xenon Tetrafluoride Molecule - The best lewis structure for the xenon tetrafluoride molecule (xef 4) is show below. Now that we know the valence electrons of xenon tetrafluoride, it will be easier for you to draw its lewis structure. A video explanation of how to draw the lewis dot structure for xenon. This lewis dot structure is a pictorial representation of valence electrons around individual atoms in a molecule along with the bond it forms. There are 2 steps to solve this one. Lewis structures show all of the valence electrons in an atom or molecule. The valence electrons are the electrons in the. Find the total valence electrons in xef4 molecule. This problem has been solved! Here, the given molecule is xef4 (xenon tetrafluoride). Web in xef4 (xenon tetrafluoride) lewis structure, there are four sigma bonds and two lone pairs around xenon atom. Web to draw lewis structures for molecules and polyatomic ions with one central atom. (valence electrons are the number of electrons present in the outermost shell of an atom). The valence electrons are the electrons in the. Web to draw a. Now that we know the valence electrons of xenon tetrafluoride, it will be easier for you to draw its lewis structure. Is its molecular structure tetrahedral or square planar? Xenon (xe) can have more than 8 valence electrons in your lewis structure. Draw the lewis structure for the xenon tetrafluoride (xef4 molecule c x. Each fluorine atom has three lone. In this tutorial, we will learn how to draw lewis structure of xef4 step by step. In some molecules, the central atom exceeds the octet rule (is surrounded by more than eight electrons). 7 electrons x 4 = 28 electrons total: In order to draw the lewis structure of xef4, first of all you have to find the total number. Lewis structures show all of the valence electrons in an atom or molecule. Web xef4 lewis structure. 44k views 6 years ago. In order to draw the lewis structure of xef4, first of all you have to find the total number of valence electrons present in the xef4 molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). Hydrogen (h) only needs two valence electrons to have a full outer shell. View the full answer step 2. There are 2 steps to solve this one. What is the hybridization of the central atom? Web to draw a lewis structure we will first need to know the total number of valence electrons in xef4 which participate in the bond formation. Check out this video for knowing the total number. 120k views 12 years ago every video. Find the total valence electrons in xef4 molecule. A lewis structure is a way to show how atoms share electrons when they form a molecule. Web the lewis structure for xef 4 requires you to place more than 8 valence electrons on xe. Now that we know the valence electrons of xenon tetrafluoride, it will be easier for you to draw its lewis structure.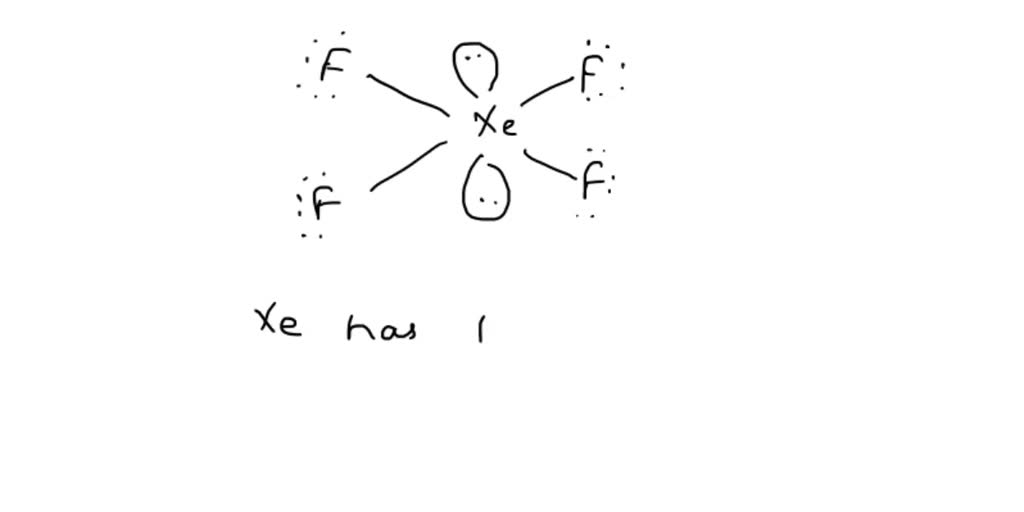
SOLVED Draw the correct Lewis structure for xenon tetrafluoride

How to Write the Formula for Xenon tetrafluoride YouTube

XeF4 Molecular Geometry, Bond Angles & Electron Geometry(Xenon
Is Its Molecular Structure Tetrahedral Or Square Planar?
Web To Draw Lewis Structures For Molecules And Polyatomic Ions With One Central Atom.
Steps Of Drawing Xef4 Lewis Structure.
Count The Total Number Of Valence Electrons.
Related Post: