Draw The Lewis Structure For Ph3
Draw The Lewis Structure For Ph3 - Lewis structure of ph3 (phosphine) chemistnate. The bond angle for the ph3 molecule. Web home | science | chemistry. Web lewis structure of ph3 contains three single bonds between the phosphorus (p) atom and each hydrogen (h) atom. A lewis structure is a way to show how atoms share. In the full lewis structure, each carbon atom satisfies the octet rule, and there are no unshared electron. Web to draw lewis structures for molecules and polyatomic ions with one central atom. 3.5k views 11 months ago. Web added jun 9, 2014 by webtester in chemistry. In the ph 3 lewis structure (and all lewis structures) hydrogen goes on the outside. Web 1) draw a lewis structure for ph3 in which the central p atom obeys the octet rule, and answer the following questions based on your drawing. Therefore, the trial structure is the correct lewis structure. Web lewis structure of phosphene. Identify the number of valence electrons for each atom in the. In the full lewis structure, each carbon atom. Total valence electrons pairs around phosphorous atom is four. Web added jun 9, 2014 by webtester in chemistry. The bond angle for the ph3 molecule. Molecular geometry around phosphorous atom is tetrahedral. Lewis structure of ph3 (phosphine) chemistnate. Web the partial lewis structure that follows is for a hydrocarbon molecule. The trial structure has exactly the same number of electrons as we have available. A video explanation of how to draw the lewis dot structure for. The number of unshared pairs. Web home | science | chemistry. Web the lewis structure for ph 3 is similar to nh 3. Find the total valence electrons for the ph3 molecule. The bond angle for the ph3 molecule. Web shape of ph 3 is trigonal pyramidal. Therefore, the trial structure is the correct lewis structure. A video explanation of how to draw the lewis dot structure for. Web lewis structure of phosphene. * phosphorus requires a full octet of. Web home | science | chemistry. 49k views 11 years ago chemistry lewis dot structures. Web phosphorus atom is in the centre forming single bonds with three hydrogen atoms and also has a lone pair of electrons in its lewis structure. Web to draw lewis structures for molecules and polyatomic ions with one central atom. To use lewis dot symbols to explain the stoichiometry of a compound. Web added jun 9, 2014 by webtester in chemistry. Remember, too, that hydrogen only needs two. Send feedback | visit wolfram|alpha.
PH3 Lewis Structure How to Draw the Lewis Structure for PH3 YouTube

Step2 Lewis Structure of PH3 for constructing around the central
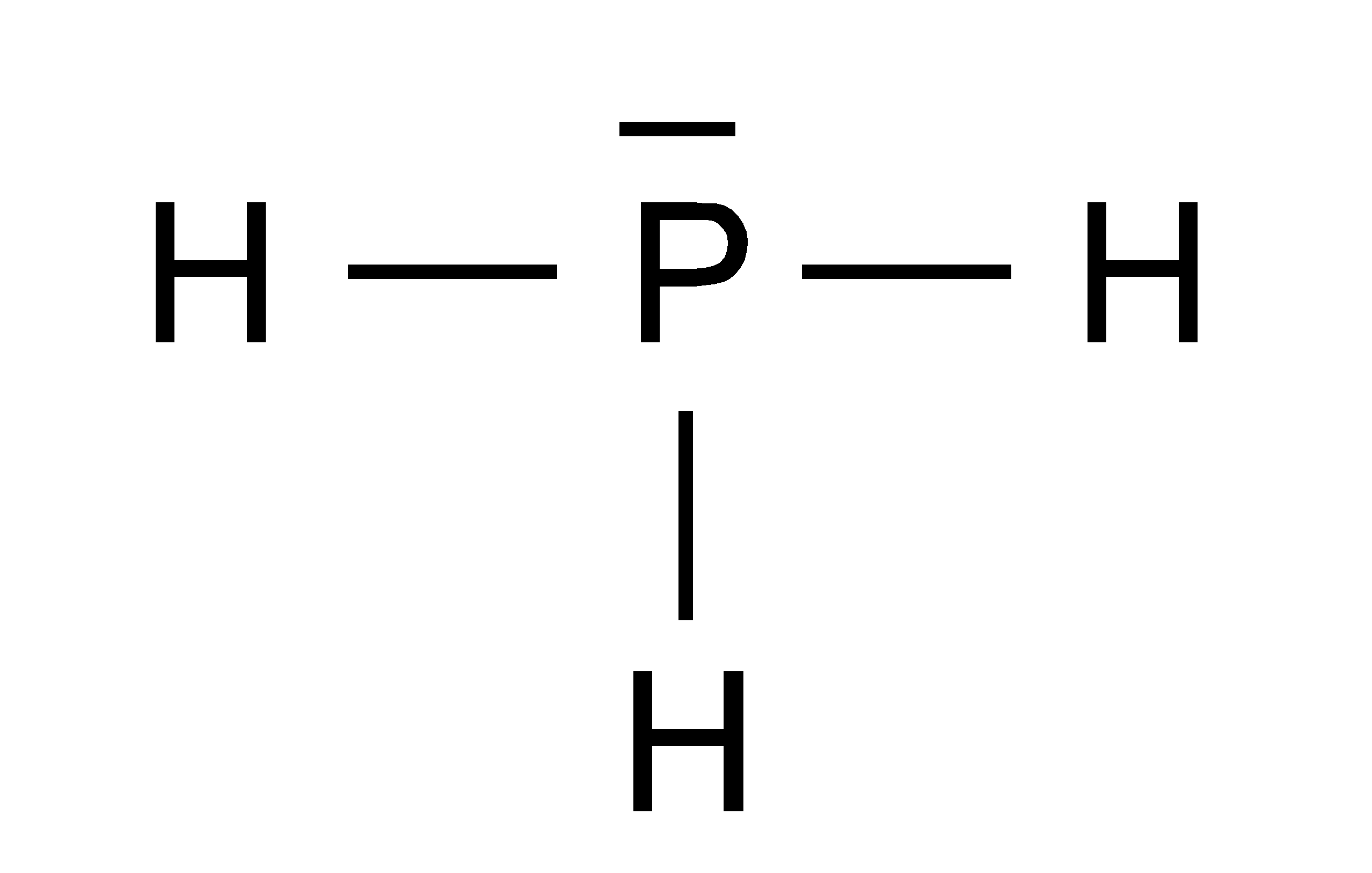
Ph3 Lewis Structure Shape
Drawings, Hybridization, Shape, Charges, Pair And Detailed Facts.
Total Valence Electrons Pairs Around Phosphorous Atom Is Four.
We Draw Lewis Structures To Predict:
Web The Partial Lewis Structure That Follows Is For A Hydrocarbon Molecule.
Related Post: