Draw Lewis Structure Co
Draw Lewis Structure Co - How can i draw a lewis structure of a compound? Using lewis structures to show valence electrons. In order to draw the lewis structure of co, first of all you have to find the total number of valence electrons present in the co molecule. Here, the given molecule is co (carbon monoxide). That will normally be the least electronegative atom (c). Find more chemistry widgets in wolfram|alpha. Web the lewis structure of carbon monoxide (co) has a triple bond formation where one is strong sigma, and the other two are weak pi bonds. Web there are guidelines (several steps) to draw a lewis structure. Draw lewis structures depicting the bonding in simple molecules. The reason for learning to draw lewis structures is to predict the number and type of bonds that may be formed around an atom. Write lewis symbols for neutral atoms and ions. Web what is the lewis structure of carbon monoxide co? Number of steps can be changed according the complexity of the molecule or ion. Count the total number of valence electrons of carbon and oxygen atom. Basis on the periodic table, lewis structure has 4 for carbon and 6 for oxygen. Using formal charges to determine how many bonds to make, a different perspective. (valence electrons are the number of electrons present in the outermost shell of an atom). Web drawing the co lewis structure involves several steps: Web here are the steps that i follow when drawing a lewis structure. The example is for the nitrate ion. This is the co lewis structure: A lewis structure also helps to make a prediction about the geometry of a molecule. How to draw the lewis dot diagram for carbon monoxide (co) it is helpful if you: By the end of this section, you will be able to: Web the lewis structure of carbon monoxide (co) has a triple bond. Connect the atoms with single bonds; Co formed by adding carbon and oxygen. Web we are required to draw the lewis structure of the molecule having the chemical formula c o Write lewis symbols for neutral atoms and ions. Try to draw the co lewis structure before watching the video. Thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions. 125k views 3 years ago. Web the lewis structure (lewis dot diagram) for co. Oxygen is present in group 16. A lewis structure is a diagram that shows the chemical bonds between atoms in a molecule and the valence electrons or lone pairs of electrons. Carbon is present in group 14. This is the co lewis structure: How to draw a lewis structure. Web for the co lewis structure you'll need a triple bond between the carbon and oxygen atoms in order to satisfy the octets of each atom while still using the 10 valence electrons available for the co molecule. Web drawing lewis structures for molecules with one central atom: Here, the given molecule is co (carbon monoxide).
Lewis Dot Structure Definition, Examples, and Drawing

Carbon Monoxide Lewis Structure
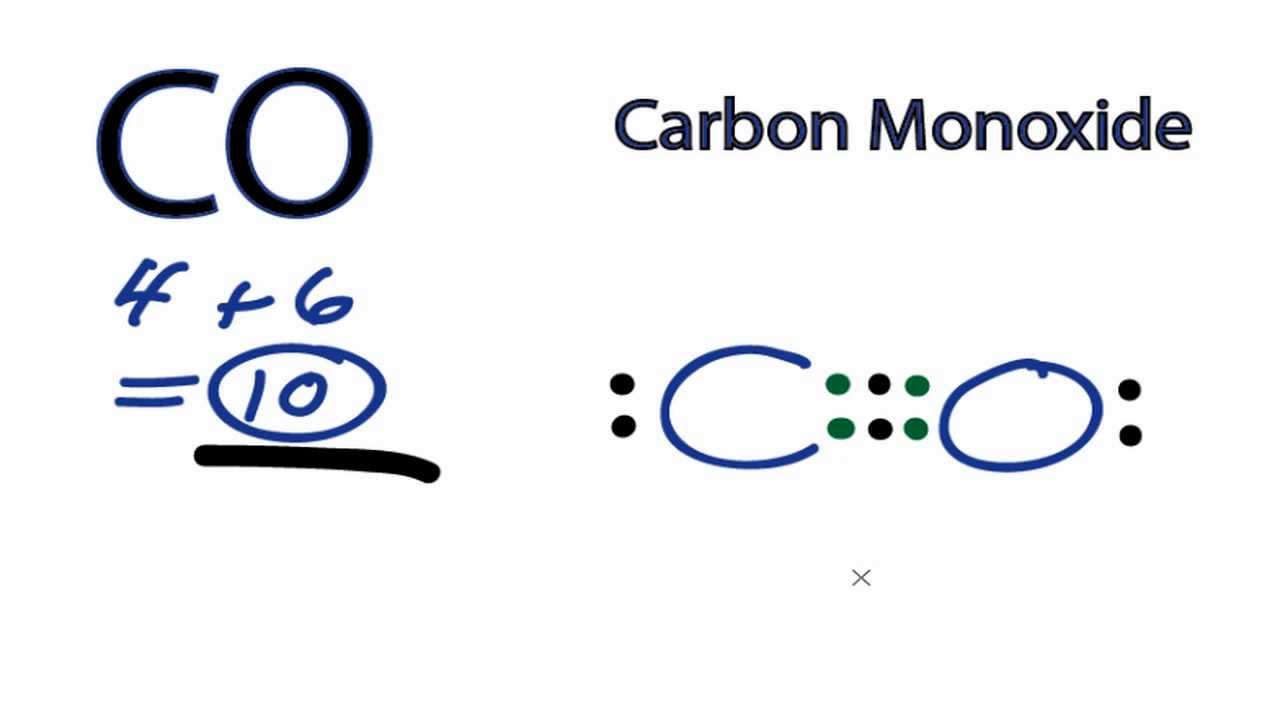
Carbon Monoxide Lewis Structure
Determine The Total Number Of Valence Electrons;
The Electronic Configuration Of Both ,O = 1S2 2S2 2P4 (6 Valence Electrons) C = 1S2 2S2 2P2 (4 Valence Electrons).
Decide Which Is The Central Atom In The Structure.
Using The Periodic Table To Draw Lewis Dot Structures.
Related Post: