Activation Energy Drawing
Activation Energy Drawing - Web in the arrhenius model of reaction rates, activation energy is the minimum amount of energy that must be available to reactants for a chemical reaction to occur. Web the potential energy diagram can illustrate the mechanism for a reaction by showing each elementary step of the reaction with distinct activation energy (see figure below ). Determine the energies of the reactants and the products. If the reaction were to proceed in the reverse direction (endergonic), the transition state would remain the same, but. Taking log on both sides. The reactant energy is 2 kj/mol while that of the products is ~0.5 kj/mol. The greater the initial rise then the more energy that is required to get the reaction going e.g., more heat needed Web when a collides with bc, the collision has to have enough kinetic energy to overcome the activation energy necessary for this reaction to occur. Energy diagrams show the amount of relative potential energy in each step of a reaction. If you had an endothermic reaction, a. Web a minimum energy (activation energy,v\(e_a\)) is required for a collision between molecules to result in a chemical reaction. The potential energy diagram shows an activation energy peak for each of the elementary steps of the reaction. Web the potential energy diagram can illustrate the mechanism for a reaction by showing each elementary step of the reaction with distinct activation. The reactant energy is 2 kj/mol while that of the products is ~0.5 kj/mol. Graphical determination of ea example plot. The greater the initial rise then the more energy that is required to get the reaction going e.g., more heat needed Determine the energies of the reactants and the products. If the reaction were to proceed in the reverse direction. The reactant energy is 2 kj/mol while that of the products is ~0.5 kj/mol. Determine the energies of the reactants and the products. Web in the arrhenius model of reaction rates, activation energy is the minimum amount of energy that must be available to reactants for a chemical reaction to occur. Graphical determination of ea example plot. The formula used. And on this energy profile, the activation energy is the difference in energy between the reactants and the transition state, so the very peak of the hill. Plots of potential energy for a system versus the reaction coordinate show an energy barrier that must be overcome for the reaction to occur. The activation energy can also be calculated algebraically if k is known at two different temperatures: Depending on the reaction, it can also include intermediates and activation energy. The higher the energy hill, the slower the reaction. Web sketch the reaction profile (reaction energy diagram) illustrated in the simulation. Label the sketch using the following terms: Energy diagrams show the amount of relative potential energy in each step of a reaction. Taking log on both sides. Web the initial increase in energy, from the reactants to the peak of the curve, represents the activation energy, e a, required to start the reaction; In the diagram above, you can clearly see that you need an input of energy to get the reaction going. The greater the initial rise then the more energy that is required to get the reaction going e.g., more heat needed The higher the energy hill, the slower the reaction. First the bonds of the reactants are broken which requires an input of energy to be put into the reaction. Web in this video, i go over how to properly label and explain a reaction mechanism diagram which is also referred to as an energy diagram or energy graph. The reactants, transition states, and products.
What Is Activation Energy? Definition and Examples
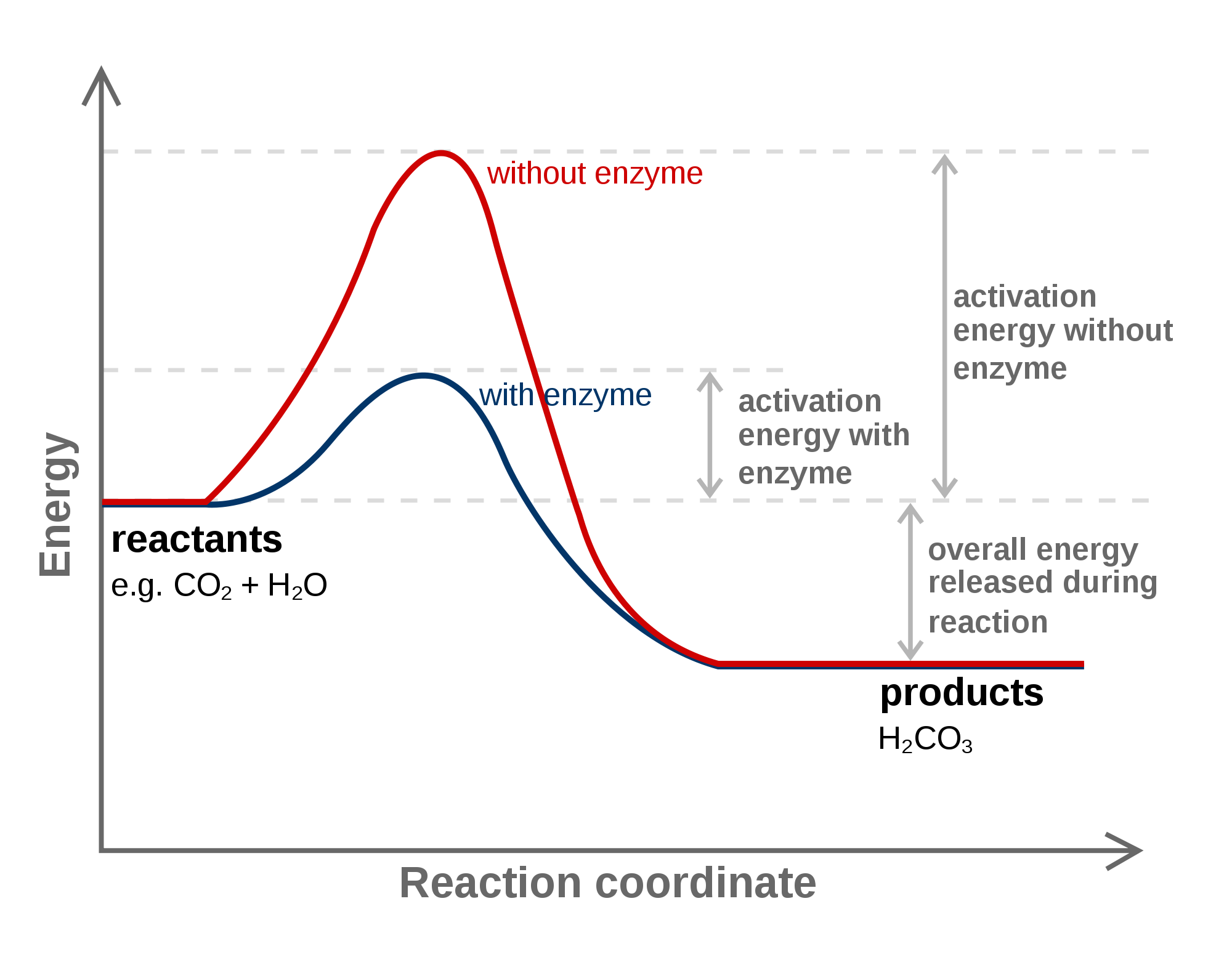
What are activation energies? Socratic
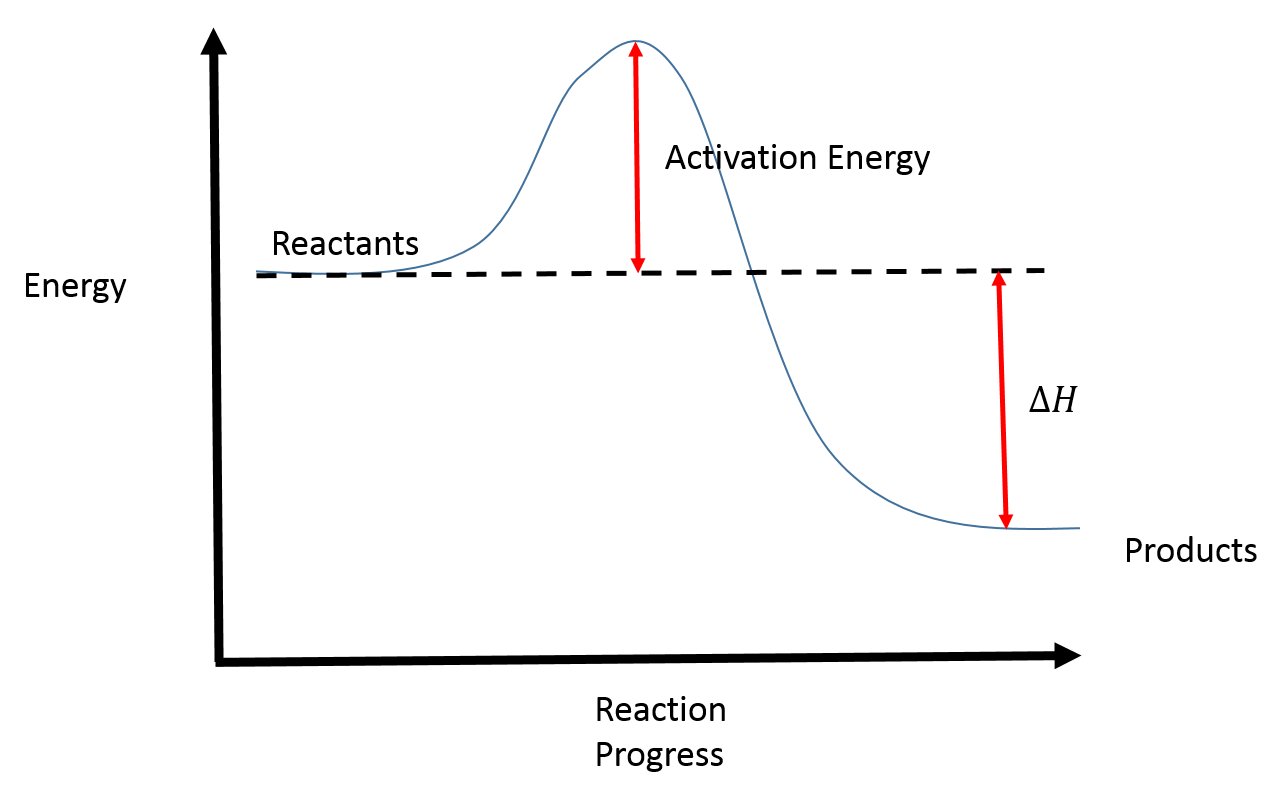
Reaction Coordinate Diagrams College Chemistry
This Type Of Diagram Shows How Energy Changes During The Course Of A.
Web In The Arrhenius Model Of Reaction Rates, Activation Energy Is The Minimum Amount Of Energy That Must Be Available To Reactants For A Chemical Reaction To Occur.
For Example, You May Want To Know What Energy Is Needed To Light A Match.
The Potential Energy Diagram Shows An Activation Energy Peak For Each Of The Elementary Steps Of The Reaction.
Related Post: